Although hydrogen produced from renewable energies has been under discussion for decades as a possible alternative to fossil fuels, it has so far only played a minor role. Recently, however, there have been signs of change, so that “green” hydrogen could gain momentum in the energy sector: More and more powerful electrolysis systems are available, and the prices for these systems are falling. If, however, PEM electrolysers were to be added on a large scale, iridium could become scarce and thus more expensive and thus stand in the way of a reduction in the already considerable investment costs.
At present, iridium-based catalysts are not yet particularly relevant for total costs, but this could change soon. Therefore, in this article we would like to highlight the role of the raw material iridium, which is critical for PEM electrolysis, and show a possible solution against scarcity.
PEM electrolysis with its high dynamics, its outstanding hydrogen quality without complex post-treatment steps, its very high power density and the already high technological maturity represents a particularly promising technology for H2 production. A major boost is also currently coming from increasingly cheaper renewable electricity generation capacities. Low electricity prices are essential for the competitive electrolytic production of hydrogen. Hydrogen from the classic steam reforming of natural gas currently costs about 2 €/kg including investment, operation and maintenance of the plants [1]. If the H2 gas were to be produced electrolytically with a system efficiency of 70 percent (Lower Heating Value – LHV), electricity costs of 4 Ct/kWh would already result in 2 €/kg operating costs (neglecting all other costs incurred in purchasing and operating the electrolyser). In order to be on a par with conventional fossil fuel production, the total costs for electrolytic hydrogen must be reduced even further.
Iridium in PEM electrolysis
Water splitting during electrolysis takes place at the anode and is a relatively slow reaction. For each molecule to be cleaved, four electrons must be transferred during this oxygen evolution reaction (OER). Accordingly, a highly active catalyst is important in order to keep overvoltage and thus losses to a minimum.
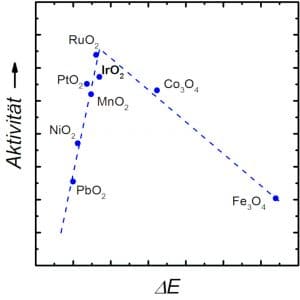
Figure 1 shows in the so-called Vulcano plot the activity of different materials for the OER over the affinity of the products and educts of the reaction to the material surface: Neither should the surface of the catalyst allow the water to adhere poorly (left area), nor should the products O2 and H+ only be detached hesitantly (right area). Thus, the materials near the tip of the Vulcano plot in particular are theoretically well suited for use as OER catalysts. In real operation, however, the stability of the materials is another decisive aspect.
…
read more in H2-international July 2019
Authors:
Maximilian Möckl, Dr. Maximilian Bernt,
both Bayerisches Zentrum für Angewandte Energieforschung e.V. (ZAE Bayern), Garching























0 Comments